Background: Often a regulatory requirement, Anti-Drug Antibody (ADA) assays are a critical component to assessing immunogenicity of a novel therapeutic. The myriad of immune responses that a novel therapeutic can induce, including generation of antibodies, can play a substantial role in affecting pharmacokinetics, pharmacodynamics, and determining the safety and efficacy of the therapeutic. As such, monitoring ADA responses during a clinical trial can provide valuable insight into the body’s immune response, and guide critical decisions on safety; especially for first-in-human trials.
Principle: ADA assays can be performed using several immunoassay formats, but the objective is to capture IgG, IgM, or IgE ADA’s from test subject sera. Detection is then performed via a secondary antibody (anti- IgG, IgM, IgE). Below is an example of how this is performed in the laboratory.
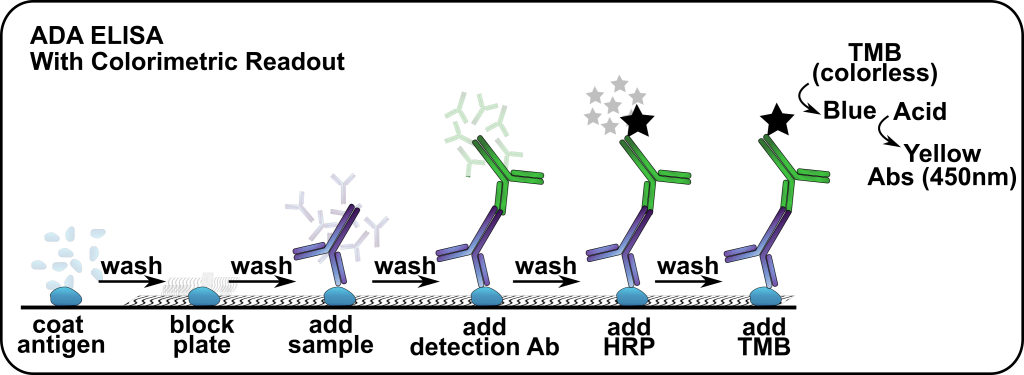
Following FDA guidance, ADA assays take a risk-based and tiered approach to evaluating immune responses. Primarily, ADA assays are divided into four phases: screening, confirmatory, titer, and neutralizing. The first two phases are designed to rule out any false-positives; then when a positive is detected the following phases are utilized to determine the amount of ADA and to characterize the ADA’s ability to interfere (neutralize) the therapeutic .
Our Approach: At Xeno Diagnostics, ADA assays are tailored for each different therapeutic. We leverage our assay development expertise to ensure that every condition from buffers, blockers, and more are tailored to maximize binding kinetics for your product while minimizing errant noise.
ADA assays are validated to GLP regulations, and clients are informed at each stage of the process: from assay development and validation through statistical analysis and cut-point determination. Our Assay Development and Validation services are designed to match the regulatory requirement, scientific goal, and budget of each client.
References:
Related Topics and Assays:
"*" indicates required fields
Copyright © 2021. All rights reserved.